Increasing Electrocatalytic Nitrate Reduction Activity by Controlling Adsorption through PtRu Alloying
11 Aug 2020·
 ,
,
·
1 min read
,
,
·
1 min read
Zixuan Wang
Samuel D. Young
Bryan Goldsmith
Nirala Singh
Abstract
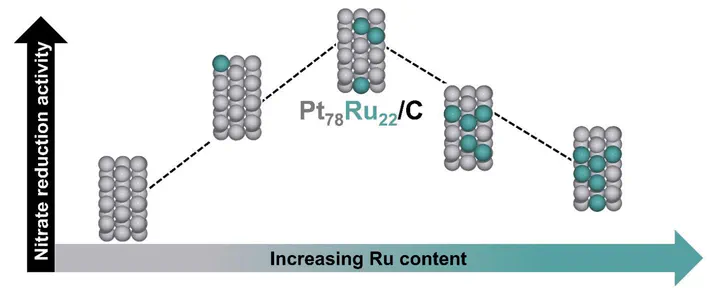
Nitrate produced from industrial and agricultural processes has imbalanced the global nitrogen cycle. Electrocatalytic reduction is a sustainable route to remediate nitrate while generating products such as ammonia or N2. Here we report the surface-area normalized activity of platinum-ruthenium (PtxRuy/C) catalysts of different compositions (x = 48–100%) for electrocatalytic nitrate reduction, chosen based on screening using a computational activity volcano plot. The PtxRuy/C alloys are more active than Pt/C, with Pt78Ru22/C six times more active than Pt/C at 0.1 V vs. RHE, and ammonia faradaic efficiencies of 93–98%. Density functional theory calculations predict maximum activity at 25 at% Ru, consistent with experiments. This maximum is due to a transition from nitrate dissociation as the rate determining step to a new rate determining step at higher Ru content. This study demonstrates how electrocatalyst performance is tunable by changing the adsorption strength of reacting species through alloying.
Type
Publication
Journal of Catalysis
Supporting information available here.